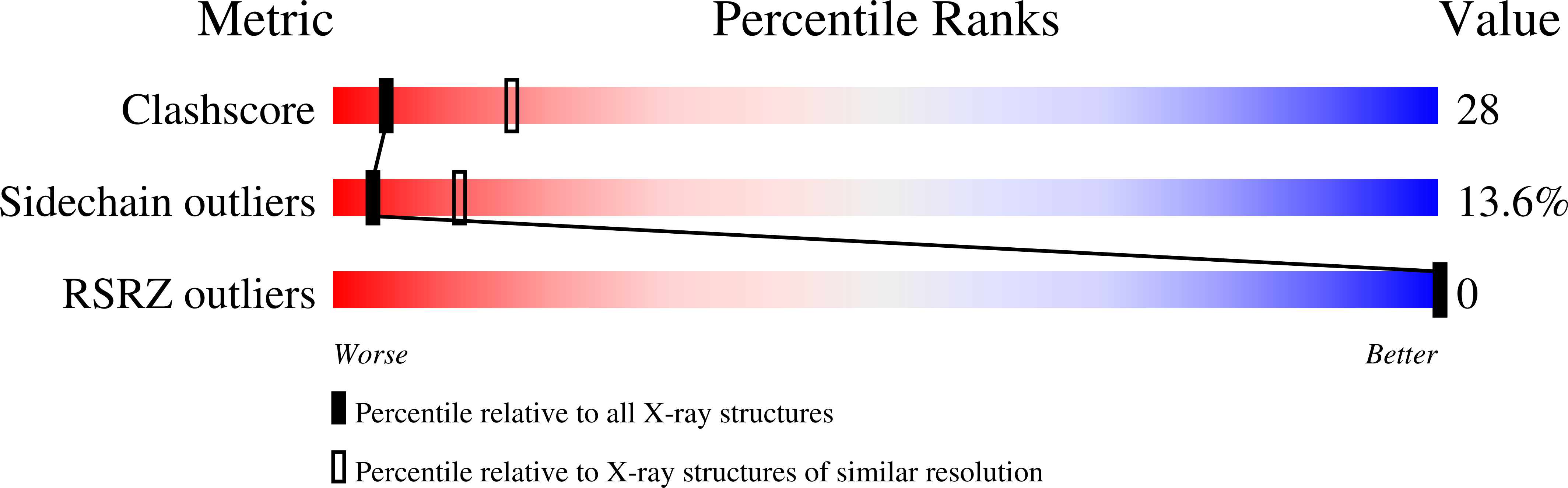Role of the N-Terminal Extension of the (Betaalpha)(8)-Barrel Enzyme Indole-3-Glycerol Phosphate Synthase for its Fold, Stability, and Catalytic Activity.
Schneider, B., Knoechel, T., Darimont, B., Hennig, M., Dietrich, S., Babinger, K., Kirschner, K., Sterner, R.(2005) Biochemistry 44: 16405
- PubMed: 16342933
- DOI: https://doi.org/10.1021/bi051640n
- Primary Citation of Related Structures:
2C3Z - PubMed Abstract:
Indole-3-glycerol phosphate synthase (IGPS) catalyzes the fifth step in the biosynthesis of tryptophan. It belongs to the large and versatile family of (betaalpha)(8)-barrel enzymes but has an unusual N-terminal extension of about 40 residues. Limited proteolysis with trypsin of IGPS from both Sulfolobus solfataricus (sIGPS) and Thermotoga maritima (tIGPS) removes about 25 N-terminal residues and one of the two extra helices contained therein. To assess the role of the extension, the N-terminally truncated variants sIGPSDelta(1-26) and tIGPSDelta(1-25) were produced recombinantly in Escherichia coli, purified, and characterized in comparison to the wild-type enzymes. Both sIGPSDelta(1-26) and tIGPSDelta(1-25) have unchanged oligomerization states and turnover numbers. In contrast, their Michaelis constants for the substrate 1-(o-carboxyphenylamino)-1-deoxyribulose 5-phosphate are increased, and their resistance toward unfolding induced by heat and guanidinium chloride is decreased. sIGPSDelta(1-26) was crystallized, and its X-ray structure was solved at 2.8 A resolution. The comparison with the known structure of sIGPS reveals small differences that account for its reduced substrate affinity and protein stability. The structure of the core of sIGPSDelta(1-26) is, however, unchanged compared to sIGPS, explaining its retained catalytic activity and consistent with the idea that it evolved from the same ancestor as the phosphoribosyl anthranilate isomerase and the alpha-subunit of tryptophan synthase. These (betaalpha)(8)-barrel enzymes catalyze the reactions preceding and following IGPS in tryptophan biosynthesis but lack an N-terminal extension.
Organizational Affiliation:
Institut für Biophysik und physikalische Biochemie, Universität Regensburg, Universitätsstrasse 31, D-93053 Regensburg, Germany.