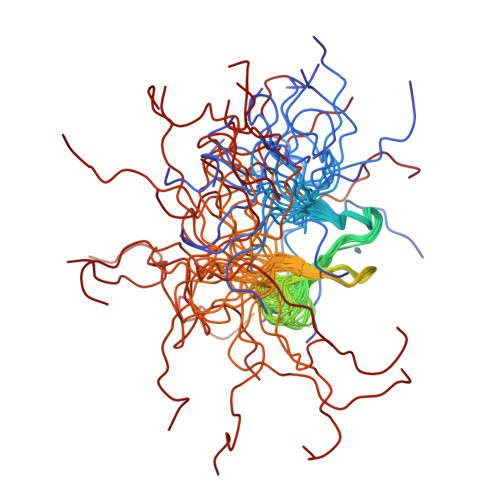
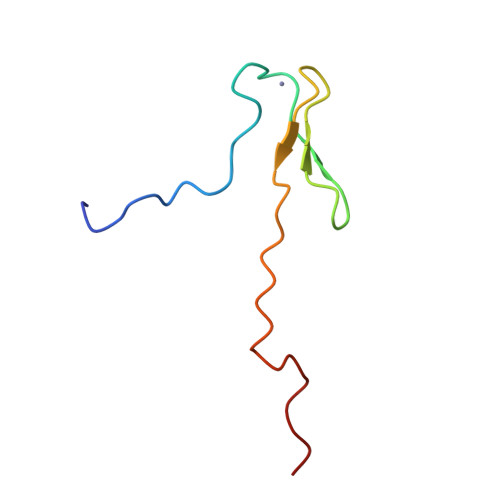
Structure of a (Cys3His) zinc ribbon, a ubiquitous motif in archaeal and eucaryal transcription.
Chen, H.T., Legault, P., Glushka, J., Omichinski, J.G., Scott, R.A.(2000) Protein Sci 9: 1743-1752
- PubMed: 11045620
- DOI: https://doi.org/10.1110/ps.9.9.1743
- Primary Citation of Related Structures:
1DL6 - PubMed Abstract:
Transcription factor IIB (TFIIB) is an essential component in the formation of the transcription initiation complex in eucaryal and archaeal transcription. TFIIB interacts with a promoter complex containing the TATA-binding protein (TBP) to facilitate interaction with RNA polymerase II (RNA pol II) and the associated transcription factor IIF (TFIIF). TFIIB contains a zinc-binding motif near the N-terminus that is directly involved in the interaction with RNA pol II/TFIIF and plays a crucial role in selecting the transcription initiation site. The solution structure of the N-terminal residues 2-59 of human TFIIB was determined by multidimensional NMR spectroscopy. The structure consists of a nearly tetrahedral Zn(Cys)3(His)1 site confined by type I and "rubredoxin" turns, three antiparallel beta-strands, and disordered loops. The structure is similar to the reported zinc-ribbon motifs in several transcription-related proteins from archaea and eucarya, including Pyrococcus furiosus transcription factor B (PfTFB), human and yeast transcription factor IIS (TFIIS), and Thermococcus celer RNA polymerase II subunit M (TcRPOM). The zinc-ribbon structure of TFIIB, in conjunction with the biochemical analyses, suggests that residues on the beta-sheet are involved in the interaction with RNA pol II/TFIIF, while the zinc-binding site may increase the stability of the beta-sheet.
Organizational Affiliation:
Center for Metalloenzyme Studies, University of Georgia, Athens 30602-2556, USA.