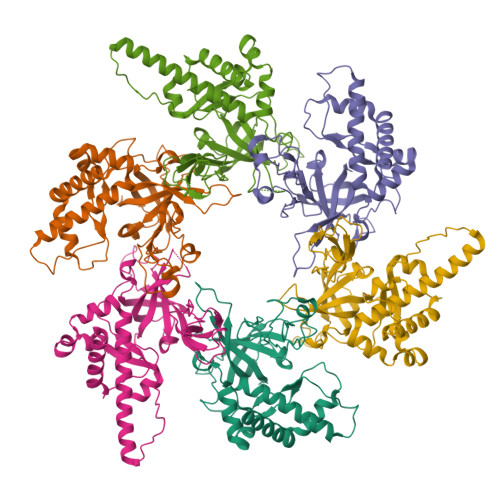
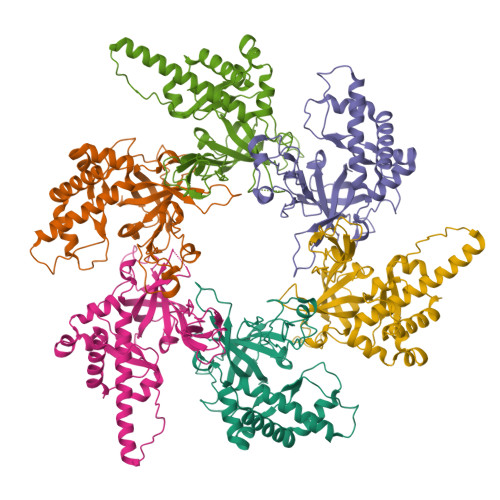
Structural and mechanistic insights into the MCM8/9 helicase complex.
Weng, Z., Zheng, J., Zhou, Y., Lu, Z., Wu, Y., Xu, D., Li, H., Liang, H., Liu, Y.(2023) Elife 12
- PubMed: 37535404
- DOI: https://doi.org/10.7554/eLife.87468
- Primary Citation of Related Structures:
7W7P, 7YOX - PubMed Abstract:
MCM8 and MCM9 form a functional helicase complex (MCM8/9) that plays an essential role in DNA homologous recombination repair for DNA double-strand break. However, the structural characterization of MCM8/9 for DNA binding/unwinding remains unclear. Here, we report structures of the MCM8/9 complex using cryo-electron microscopy single particle analysis. The structures reveal that MCM8/9 is arranged into a heterohexamer through a threefold symmetry axis, creating a central channel that accommodates DNA. Multiple characteristic hairpins from the N-terminal oligosaccharide/oligonucleotide (OB) domains of MCM8/9 protrude into the central channel and serve to unwind the duplex DNA. When activated by HROB, the structure of MCM8/9's N-tier ring converts its symmetry from C3 to C1 with a conformational change that expands the MCM8/9's trimer interface. Moreover, our structural dynamic analyses revealed that the flexible C-tier ring exhibited rotary motions relative to the N-tier ring, which is required for the unwinding ability of MCM8/9. In summary, our structural and biochemistry study provides a basis for understanding the DNA unwinding mechanism of MCM8/9 helicase in homologous recombination.
Organizational Affiliation:
Shenzhen Key Laboratory for Systems Medicine in Inflammatory Diseases, School of Medicine, Shenzhen Campus of Sun Yat-sen University, Shenzhen, China.