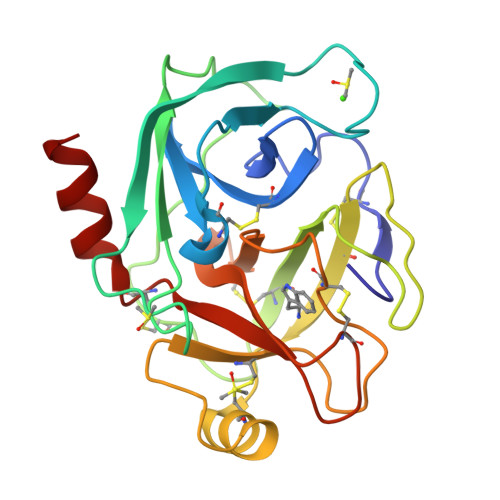
In-crystal affinity ranking of fragment hit compounds reveals a relationship with their inhibitory activities
Yamane, J., Yao, M., Zhou, Y., Hiramatsu, Y., Fujiwara, K., Yamaguchi, T., Yamaguchi, H., Togame, H., Tsujishita, H., Takemoto, H., Tanaka, I.(2011) J Appl Crystallogr 44: 798-804